1.800.858.7378npic@oregonstate.edu
Call, email, or chat Mon-Fri
A to Z

This page explains the chemical structures of quaternary ammonium compounds (quats). For information on the health effects and uses of quats, see the Quaternary Ammonium Compounds (ADBAC/DDAC) Fact Sheet.
Quaternary ammonium compounds, also called quats, are chemicals made of two basic parts. The first part is a central nitrogen atom.1 The second part is made up of four clusters of atoms2 that are attached to the nitrogen atom (Figure 1). The U.S. Environmental Protection Agency (EPA) currently classifies quats into four different groups based on the structure of the four clusters attached to the central atom. The physical and chemical properties of the quats contribute to the behavior of the quat and how toxic it is. The EPA has identified specific compounds in each group to represent the rest of them when they do toxicity testing.
Some quats may have chlorine atoms attached to them. These are called chlorides, which means the chlorine atom borrows an electron from the nitrogen atom. The chlorine atom, with its borrowed electron, has a negative electrical charge. When the nitrogen atom gives up one of its electrons, it is called an ammonium ion. It has a positive electrical charge. Overall, the compound has no electrical charge because the positive and negative charges balance out. Compounds like these are salts.
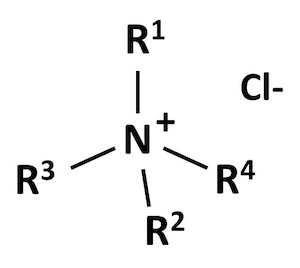
Figure 1. Quaternary ammonium cation general structure. "R" is any cluster of atoms.
Group I Quats
Group I quats are called "didecyl dimethyl ammonium chloride", or DDAC for short. These are the simplest quats that have straight chains of carbon atoms attached to the nitrogen (Figure 2). "Didecyl" means that two of the clusters are chains of carbon atoms that are at least 10 atoms long. "Dimethyl" means that the other two clusters attached to the nitrogen atom are just a single carbon atom with hydrogen atoms attached.
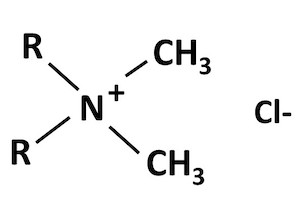
Figure 2. General structure of Group I Quats: DDAC
Group II Quats
Group II quats are called "alkyl dimethyl benzyl ammonium chloride", or ADBAC for short. These have two clusters made up of a single carbon atom each. The third cluster is a long chain of carbon atoms attached to the nitrogen atom. The fourth cluster is in the form of a ring. The carbon atoms that make up the ring have only hydrogen atoms attached to them. That ring is called a "benzyl" ring3 (Figure 3). Another name for this basic structure is benzalkonium chloride. This is sometimes written as BAC for short. The EPA has combined 24 compounds with this basic structure under Group II quats.
- The atom is the smallest unit of matter that is composed of three subatomic particles: the proton, the neutron, and the electron. Protons and neutrons make up the nucleus of the atom, a dense and positively charged core, whereas the negatively charged electrons can be found around the nucleus in an electron cloud.
- Clusters are a group of atoms, molecules, or ions that adhere together under forces like those that bind the atoms.
- Benzyl features a benzene ring (C6H6) attached to a methylene group (−CH2−) group.
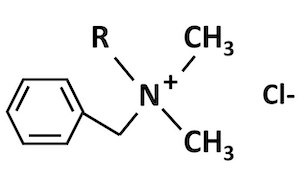
Figure 3. General structure of Group II Quats: ADBAC
Group III Quats
Group III quats are more complicated. They have two chlorine atoms stuck to the benzyl ring instead of just hydrogen atoms. These chlorine atoms are always in the same position on the ring. The EPA has allowed one particular chemical in the group to be used for all of the required toxicity testing for Group III quats.
Group IV Quats
The last group, Group IV quats, are even more complicated than Group III. Each compound in this group must undergo separate toxicity testing because these quats do not all behave in the same way. One Group IV quat's toxicity cannot predict another's.
How to Identify Quats
The names of chemicals give us hints about how they are built by describing their parts. However, there are variations in how chemists describe or name chemical compounds. There may be more than one name that describes the same compound. That's why chemists use tools like a Chemical Abstracts Service (CAS) Registry Number to identify compounds. CAS numbers are listed on Safety Data Sheets and sometimes pesticide labels. When the chemical name is long or complex, you can look up the CAS number to make sure you have the right compound. Table 1 below lists all the unique chemicals in Group 1 (DDAC) and Group II (ADBAC).
| Table 1. List of chemicals included in Group I (DDAC) and Group II (ADBAC) quaternary ammonium compounds.* The U.S. EPA assigns a pesticide code number (PC Number) to pesticide active ingredients or mixes of active ingredients. | ||
|---|---|---|
| Group I Cluster - Didecyl dimethyl ammonium chloride (DDAC) | ||
| Chemical Name | CAS Number(s) | PC Number(s) |
| Didecyl dimethyl ammonium chloride | 7173-51-5 | 69149 |
| Dioctyl dimethyl ammonium chloride | 5538-94-3 | 69166 |
| Octyl decyl dimethyl ammonium chloride | 32426-11-2 | 69165 |
| Alkyl dimethyl ethyl ammonium bromide | 84540-07-8 | 69146 |
| Oxydiethylenebis (alkyl*) dimethyl ammonium chloride | 68607-28-3 | 69173 |
| Group II Cluster – Alkyl dimethyl benzyl ammonium chloride (ADBAC) | ||
| Chemical Name | CAS Number(s) | PC Number(s) |
| Alkyl dimethyl benzyl ammonium chloride |
53516-76-0 68424-85-1 8001-54-5 139-08-2 68424-85-1 61789-71-7 68424-85-1 68424-85-1 68391-01-5 68391-01-5 68424-85-1 85409-22-9 68424-85-1 63449-41-2 |
69104 69105 69106 69107 69137 69140 69141 69157 69175 69189 69184 69192 69194 128928 |
| Dialkyl methyl benzyl ammonium chloride | 73049-75-9 | 69119 |
| Alykyl dimethyl benzyl ammonium saccharinate | 68989-01-5 | 69171 |
| Alkyl dimethyl ethylbenzyl ammonium chloride | 85409-23-0 8045-21-4 |
69154 69111 |
| n-alkyl dimethyl 1-naphtylmethyl ammonium chloride | 53516-75-9 | 69112 |
| Dodecyl benzyl trimethyl ammonium chloride | 1330-85-4 | 69125 |
| n-alkyl dimethyl dimethyl ammonium chloride | N/A | 169159 |
| Diisobutylphenoxy ethyoxyethyl dimethyl benzyl ammonium chloride [Benzethonium Chloride] | 121-54-0 | 69122 |
| Methyl dodecyl benzyl tri methyl ammonium chloride - 80% Methyl dodecyl xylene bis tri methyl ammonium chloride – 20% | 1399-80-0 | 69129 |
|
*Sources:
|
||
Additional Resources:
- DDAC Reregistration Eligibility Decision - U.S. EPA
- ADBAC Reregistration Eligibility Decision - U.S. EPA
- Quaternary Ammonium Compounds (QACs) - Toxics Use Reduction Institute, U Mass, Lowell
- Selected EPA-Registered Disinfectants - U.S. EPA
- What's the difference between products that disinfect, sanitize, and clean surfaces? - U.S. EPA
- Characteristics of Selected Disinfectants - Center for Food Security & Public Health, Iowa State University
If you have questions about this, or any pesticide-related topic, please call NPIC at 800-858-7378, or email us at npic@ace.orst.edu.
