1.800.858.7378npic@oregonstate.edu
Call, email, or chat Mon-Fri
A to Z
DEET Technical Fact Sheet
As of 2011, NPIC stopped creating technical pesticide fact sheets. The old collection of technical fact sheets will remain available in this archive, but they may contain out-of-date material. NPIC no longer has the capacity to consistently update them. To visit our general fact sheets, click here. For up-to-date technical fact sheets, please visit the Environmental Protection Agency’s webpage.
- Chemical Class and Type
- Physical / Chemical Properties
- Uses
- Mode of Action
- Toxicity Classification
- Acute Toxicity
- Chronic Toxicity
- Endocrine Disruption
- Carcinogenicity
- Reproductive and Teratogenic Effects
- Fate in the Body
- Medical Tests and Monitoring
- Environmental Fate
- Ecotoxicity Studies
- Regulatory Guidelines
Chemical Class and Type:
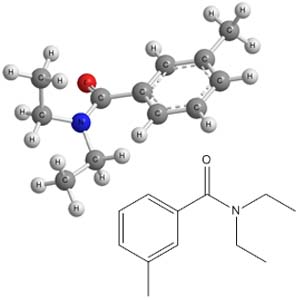
- DEET is an insect and acarid repellent. The International Union of Pure and Applied Chemistry (IUPAC) name for DEET is N,N-diethyl-m-toluamide and other isomers, and it is a member of the N,N-dialkylamide family of chemicals. The Chemical Abstracts Service (CAS) registry number for DEET is 134-62-3.1
- DEET was developed by the U.S. Army in 1946 for protection of military personnel in insect-infested areas. DEET was registered in the United States for use by the general public in 1957, and has been commercially marketed as a personal insect repellent since 1965. A Registration Standard was established for DEET by the United States Environmental Protection Agency (U.S. EPA) in 1980. DEET was reregistered in 1998 following a review of the Registration Standard and additional animal and avian toxicity data from a Data Call-In (DCI) in 1988.1 See the text box on Laboratory Testing.
Molecular Structure -
DEET
Laboratory Testing: Before pesticides are registered by the U.S. EPA, they must undergo laboratory testing for short-term (acute) and long-term (chronic) health effects. Laboratory animals are purposely given high enough doses to cause toxic effects. These tests help scientists judge how these chemicals might affect humans, domestic animals, and wildlife in cases of overexposure.
Physical / Chemical Properties:
- DEET is a liquid with a distinct, faint odor and is almost colorless.1
- Vapor pressure2: 5.6 x 10-3 mmHg at 20 °C
- Octanol-Water Partition Coefficient (log Kow)2: 2.02
- Henry's constant2: 2.1 x 10-8 atm·m3/mol
- Molecular weight2: 191.3 g/mol
- Solubility (water)1: >1.0 g/L (>1000 mg/L) at 25 °C
- Soil Sorption Coefficient (Koc)2: 3.00 x 102
- DEET is a plasticizer and can damage certain rubber, plastic, vinyl, or elastic materials such as contact lenses, eyeglass frames and lenses, watch crystals, combs, painted and varnished surfaces, and certain synthetic or treated fabrics. DEET does not damage natural fibers including cotton and wool.3
Uses:
- DEET is used to repel biting and flying insects such as biting flies and midges, black flies, chiggers, no-see-ums, fleas, mosquitoes, and ticks. Repellents containing DEET are used on a variety of sites, including the human body, clothing, headgear, and horses. Formulations of the repellent include liquids, pressurized liquids, lotions, sticks, foams, and impregnated materials such as towelettes.1,4 Uses for individual products containing DEET vary widely. Always read and follow the label when applying pesticide products.
- It has been previously estimated that approximately 30% of the U.S. population uses DEET annually as a personal insect repellent.1
- Signal words for products containing DEET may range from Caution to Danger. The signal word reflects the combined toxicity of the active ingredient and other ingredients in the product. See the pesticide label on the product and refer to the NPIC fact sheets on Signal Words and Inert or "Other" Ingredients.
- To find a list of products containing DEET which are registered in your state, visit the website https://npic.orst.edu/reg/state_agencies.html select your state then click on the link for "State Products."
- Sunscreen: The U.S. EPA is reviewing additional information regarding the use of DEET in combination with sunscreen. Instructions to apply sunscreen "generously and frequently" are in contrast to the U.S. EPA's recommendation to use DEET "sparingly".5
Mode of Action:
Target Organisms
- Scientists have not definitively determined the exact mode of action of DEET in target organisms. Research indicates that DEET interferes with receptors in mosquito (and other biting insects) antennae that detect L-lactic acid and carbon dioxide, the primary attractants emitted by humans and other animals. Insects exposed to DEET are thereby unable to effectively locate a host.6,7,8
Non-target Organisms
- Scientists have not determined the exact mechanism by which DEET may affect the nervous system in humans.9
Acute Toxicity:
Oral
- DEET is low in toxicity to rats when ingested. The acute oral LD50 in rats is 2170 to 3664 mg/kg.1 See the text boxes on Toxicity Classification and LD50/LC50.
Dermal
- DEET is low in toxicity to rabbits when applied to the skin. The acute dermal LD50 in rabbits is 4280 mg/kg.1
- Researchers observed slight to moderate dermal irritation and slight edema in studies exposing rabbits to technical grade DEET. Dermal irritation resolved within seven days.10
- Investigators did not observe skin sensitization in guinea pigs or humans treated with technical grade or formulated DEET.10
- Researchers observed slight to moderate corneal opacity, slight iritis, and moderate to severe conjunctivitis following 24- hour ocular exposure studies in rabbits with technical grade DEET. Symptoms generally cleared within five to seven days. Conjunctivitis persisted beyond seven days in some studies.10
LD50/LC50: A common measure of acute toxicity is the lethal dose (LD50) or lethal concentration (LC50) that causes death (resulting from a single or limited exposure) in 50 percent of the treated animals. LD50 is generally expressed as the dose in milligrams (mg) of chemical per kilogram (kg) of body weight. LC50 is often expressed as mg of chemical per volume (e.g., liter (L)) of medium (i.e., air or water) the organism is exposed to. Chemicals are considered highly toxic when the LD50/LC50 is small and practically non-toxic when the value is large. However, the LD50/LC50 does not reflect any effects from long-term exposure (i.e., cancer, birth defects or reproductive toxicity) that may occur at levels below those that cause death.
Inhalation
- DEET is very low in toxicity to rats when inhaled. The acute inhalation LC50 in rats is 5.95 mg/L.1
Signs of Toxicity - Animals
- Reports of DEET toxicosis in animals are rare. Clinical signs in dogs and cats may include vomiting, tremors, excitation, ataxia, and seizures.11
Signs of Toxicity - Humans
- Poison control center data from 1993-1997 revealed that symptoms were dependent on the pathway of exposure. Reported symptoms were most frequently associated with ocular exposure, with effects including eye irritation or pain, and excess tearing.12
- Dermal symptoms were reported in 5% of reports to poison control centers following DEET exposure. Symptoms included irritation, redness, rash, and swelling.12
- Gastrointestinal symptoms reported to poison control centers following exposure to DEET primarily occurred following ingestion. Gastrointestinal symptoms included oral irritation, nausea, and vomiting.12
- Neurological reactions from DEET exposure are rare. The incidence of seizure following DEET exposure is estimated to be 1 per 100 million users between 1961 and 1998. Reports of neurological reactions are predominately the result of accidental or deliberate ingestion, or dermal applications not consistent with label directions. Reported neurological signs of toxicity include encephalopathy, seizure, tremor, slurred speech, coma, and death.1,5,13
- The U.S. EPA and FIFRA Scientific Advisory Panel concluded that the reported incidences of seizures among children that were potentially related to DEET use are "inconclusive" and do not support a causal link between DEET and seizures in children.1
- Always follow label instructions and take steps to minimize exposure. If any exposure occurs, be sure to follow the First Aid instructions on the product label carefully. For additional treatment advice, contact the Poison Control Center at 1-800- 222-1222. If you wish to discuss an incident with the National Pesticide Information Center, please call 1-800-858-7378.
Chronic Toxicity:
Animals
Oral
NOAEL: No Observable Adverse Effect Level
NOEL: No Observed Effect Level
LOAEL: Lowest Observable Adverse Effect Level
LOEL: Lowest Observed Effect Level
- Researchers administered DEET in the diet of male and female rats at doses of 10, 30, or 100 mg/kg/day for males, and 30, 100 or 400 mg/kg/day for females for 2 years. Researchers observed no toxicity among males at any dose level, and observed decreased body weight, decreased food consumption, and increased cholesterol levels among females at the highest dose tested. The NOELs for oral toxicity are 100 and 400 mg/kg/day for male and female rats, respectively.1 See the text box on NOAEL, NOEL, LOAEL, and LOEL.
- Researchers administered DEET in the form of gel capsules to dogs at doses of 0, 30, 100, and 400 mg/kg/day for one year. At the highest dose tested, investigators noted decreased body weight, and increased incidence of drooling/salivation, and decreased cholesterol levels.1
Dermal
- Investigators applied DEET to the shaved backs of male and female rats at doses of 0, 100, 300, or 1000 mg/kg/day for 90 days. Researchers observed no effect on general behavior among any treatment group. Dermal irritation presented as red and scabbed areas of the skin for all treatment groups. Researchers also observed decreased body weights of male rats at the highest dose tested. The systemic NOEL determined from this study was 300 mg/kg/day for decreased body weights, and 100 mg/kg/day for local effects based on dermal irritation.10
- Researchers applied DEET to the skin of micropigs® at doses of 0, 100, 300, or 1000 mg/kg/day, five days per week for 13 weeks. Investigators observed skin effects, including dryness and desquamation at the site of application for all doses. Severity of skin effects increased with increased dose level. Investigators did not observe systemic effects at any dose tested. The LOEL for skin irritation was 100 mg/kg, and the NOEL for systemic toxicity was determined to be equal to or greater than 1000 mg/kg/day.1,10
Humans
- No data were found on chronic health effects from DEET exposure in humans. See the text box on Exposure.
Exposure: Effects of DEET on human health and the environment depend on how much DEET is present and the length and frequency of exposure. Effects also depend on the health of a person and/or certain environmental factors.
Endocrine Disruption:
- No data were found on endocrine effects from DEET exposure.
Carcinogenicity:
Animals
- Researchers fed DEET to male and female rats in the diet for two years at doses of 10, 30, or 100 mg/kg/day, and 30, 100, or 400 mg/kg/day, respectively. Researchers fed mice 250, 500, or 1,000 mg/kg/day for 18 months, and dogs 30, 100, or 400 mg/kg/day. No specific target organ toxicity or oncogenicity was observed in any of the animals.14
- Researchers often use studies designed to test for mutagenicity to screen chemicals for carcinogenicity. Sufficient evidence indicates that DEET does not have significant potential for mutagenicity.15
Humans
- The U.S. EPA classified DEET as "Group D - not classifiable as to human carcinogenicity" based on inadequate evidence of carcinogenicity, or lack of data in laboratory animals and humans.1 See the text box on Cancer.
Cancer: Government agencies in the United States and abroad have developed programs to evaluate the potential for a chemical to cause cancer. Testing guidelines and classification systems vary. To learn more about the meaning of various cancer classification descriptors listed in this fact sheet, please visit the appropriate reference, or call NPIC.
Reproductive or Teratogenic Effects:
Animals
- In a two-generation study, researchers fed rats 0, 25, 100, or 250 mg/kg/day of DEET in the diet. There were no adverse reproductive effects on fertility, gestation, or viability. At the highest dose tested, male pups exhibited reduced body weight at day 7 of lactation. Female pups experienced a significant reduction in body weight at 100 mg/kg/day at day 14 of lactation. The NOEL for reproductive toxicity was 250 mg/kg/day.1
- Researchers applied DEET at 1000 mg/kg/day to the skin of rats throughout pregnancy. Prenatal mortality increased relative to the control (34.1% vs. 20.9%). Mortality between birth and weaning increased relative to the control (44.0% vs. 15.7%).16
- Researchers administered DEET to pregnant female rabbits (route of exposure not specified) at doses of 0, 30, 100 or 325 mg/kg/day from days 6 through 18 of gestation. Scientists did not observe any compound-related maternal toxicity or developmental toxicity. The NOEL for maternal and development toxicity was 325 mg/kg/day.1
Humans
- In a double-blind, randomized trial of the safety of DEET to prevent malaria during pregnancy, females in the second and third trimester of pregnancy applied 20% DEET in carrier solution, or carrier solution alone to exposed areas of the arms and legs daily until delivery. DEET crossed the placental barrier and was detected in 8% of cord blood samples. No significant adverse effects were detected in either the mother or the fetus during pregnancy, and infants showed no differences in development or survival at birth or at age one.14
Fate in the Body:
Absorption
Animals
- Researchers fed DEET to rats and found peak blood concentrations occurring at 0.5 hours following exposure for males and at 2.0 hours following exposure for females. Male and female rats absorbed 53.3% and 65.3% of the administered dose, respectively.1
- Researchers dermally applied DEET to rats and found that blood levels reached a plateau 1.5 hours after application, and remained at that level for 24 hours (until termination of study), indicating that a small amount of DEET is continuously absorbed from the site of application. Following dermal application, male and female rats absorbed 17.0% and 5.3% of the applied dose, respectively.1
- Investigators analyzed absorption rates of a commercial sunscreen products containing 9.5% DEET compared to a 20.0% DEET solution applied to mouse skin. The sunscreen product was initially absorbed faster, and at a rate 3.4 times higher, than the 20.0% DEET solution.17 Scientists treated pigs with 10% DEET alone and in combination with 5% oxybenzone sunscreen in a skin penetration study. Compared to DEET alone, the combination of DEET and sunscreen significantly increased the absorption of DEET into the skin of laboratory animals.18 See sunscreen information under Uses.
- Investigators applied DEET to the skin of rats following oral administration of ethanol at doses of 1.5, 3.0, 4.3, 6.0, or 10.0 g/ kg to observe the effect of ethanol ingestion on DEET dermal penetration. At two hours post-treatment, ingested ethanol caused a statistically significant dose-dependent increase in DEET absorption through the skin at 4.3, 6.0, and 10.0 g/kg ethanol. At 24 hours following treatment, researchers observed enhanced absorption of DEET at doses of 6.0 g/kg and 10.0 g/kg ethanol. Doses of ethanol that caused increased absorption are relevant to human levels of alcohol consumption.21
Humans
- Researchers applied technical grade DEET, and DEET formulated in a 15% ethanol solution, to the forearm skin of male human volunteers for an 8-hour exposure period. DEET was absorbed within two hours after application and absorption continued at a constant rate over the 8-hour exposure period. Researchers determined that a total of 5.6% of the dose of technical grade DEET, compared to 8.4% of the dose of DEET in ethanol solution, was absorbed through the skin.19
- In a study to determine the effect of ethanol on the skin permeation of DEET, investigators found that 10% DEET formulated with 30-45% ethanol demonstrated higher skin permeation than a solution containing only DEET.20
Distribution
Animals
- Researchers detected very small concentrations of radio-labeled DEET primarily in the liver, kidney, lung, spleen, whole blood, and carcass of experimental animals. Total radioactivity found in all tissues accounted for between 0.15% and 0.67% of the administered dose.1
Humans
- Researchers applied DEET to the forearm skin of male human volunteers for an 8-hour exposure period and found quantifiable levels in plasma for 12 hours following the initial application. DEET did not accumulate in superficial layers of human skin.19
Metabolism
Animals
- In rats, DEET is primarily metabolized via oxidation of the methyl group on the aromatic ring, which represents 50% of an administered oral dose. The other metabolic pathway of DEET (18% of the administered oral dose) is via ring methyl oxidation in combination with N-dealkylation of an ethyl substituent on the amide moiety.1,9
Humans
- Researchers applied DEET to the forearm skin of male human volunteers for an 8-hour exposure period and found that the entire absorbed dose (5.6% and 8.3% of the applied dose, depending on formulation) was metabolized prior to excretion. Researchers found six metabolites, with the two major metabolites (up to 68% of total) resulting from metabolic processes similar to those observed in rat studies.1,19
- An in vitro study with human liver microsomes determined that ring methyl hydroxylation is the major metabolic pathway for DEET in humans. This pathway is dependent on specific CYP450 isoforms, whose levels vary considerably among individuals.9,22
Excretion
Animals
- DEET is excreted primarily in the urine in laboratory animals.1
- Researchers compared the absorption and elimination of DEET in three species of animals following a single topical application. In each species, at least 75% of the absorbed dose was detected in the urine within the first day, with elimination essentially complete within 3-4 days for all animals.23
- A pharmacokinetic study in rats exposed to radio-labeled DEET monitored excretion of the compound in the urine after single and multiple oral doses, as well as a single dermal dose. Recovery rates at seven days post-exposure for the singleand multiple-oral doses were between 85-91% from the urine and 3-5% from the feces. From the dermal dose, 74.0-78.0% was recovered from the urine, 4.0-7.0% from the feces and 6.5% from the application site at seven days post-exposure.24
Humans
- Researchers applied DEET to the forearm skin of male human volunteers for an 8-hour exposure period and found that most of the absorbed dose was eliminated within 12 hours following application, and nearly all of the absorbed dose was eliminated within 24 hours following application. Essentially all of the absorbed dose (less than 10% of the applied dose) was excreted in the urine.19
Medical Tests and Monitoring:
- Methods exist for testing DEET in blood and tissue, and DEET metabolites in urine. However, these methods are not widely available.13
- Researchers analyzed urine samples from a random, representative sub-sample of 2535 participants from the 2001-2002 National Health and Nutrition Environmental Analyses Survey (NHANES) for metabolites of DEET. Ninety-five percent (95%) of the urine samples tested for DEET were below the limit of detection (0.17 μg/L).25
Environmental Fate:
Soil
- DEET is moderately mobile in soil.2
- DEET is stable to hydrolysis at soil pH levels typically found in the environment.1
- Investigators observed microbial degradation of DEET by the soil bacterium P. putida, under conditions where DEET was the sole carbon source. The resulting metabolic products are 3-methyl benzoate and diethylamine. The metabolic pathway used by P. putida is different compared to observed pathways used by other eukaryotes.26
- Researchers observed metabolism of DEET in cultures of soil fungi. C. elegans and M. ramannianus effectively metabolized DEET yielding several metabolites by proposed mechanisms of N-oxidation and N-deethylation. Metabolic products of the fungi breakdown demonstrated lower toxicity to Daphnia magna compared to the parent compound.27
Water
- DEET is practically insoluble in water.15
- DEET has been found in water where wastewater is thought to contribute to stream-flow. The median level found was 0.05 μg/L, with the highest levels (1.10 μg/L) found in streams with urban wastewater.28
Air
- DEET will exist as a vapor in ambient air and degrade via hydroxyl- radicals with an estimated half-life of 15 hours.2 See the text box on Half-life.
The "half-life" is the time required for half of the compound to break down in the environment.
1 half-life = 50% remaining
2 half-lives = 25% remaining
3 half-lives = 12% remaining
4 half-lives = 6% remaining
5 half-lives = 3% remaining
Half-lives can vary widely based on environmental factors. The amount of chemical remaining after a half-life will always depend on the amount of the chemical originally applied. It should be noted that some chemicals may degrade into compounds of toxicological significance.
Indoor
- No indoor fate data were found for DEET.
Food Residue
- No food tolerances have been established for DEET.
Ecotoxicity Studies:
Birds
- DEET is slightly toxic to birds (oral LD50 = 1375 mg/kg).1
Fish and Aquatic Life
EC50: The median effective concentration (EC50) may be reported for sublethal or ambiguously lethal effects. This measure is used in tests involving species such as aquatic invertebrates where death may be difficult to determine. This term is also used if sublethal events are being monitored.
Newman, M.C.; Unger, M.A. Fundamentals of Ecotoxicology; CRC Press, LLC.: Boca Raton, FL, 2003; p 178.
Regulatory Guidelines:
- The U.S. EPA has classified DEET as "Group D - not classifiable as to human carcinogenicity" based on inadequate evidence of carcinogenicity, or lack of data in laboratory animals and humans.1 See the text box on Cancer.
- The Centers for Disease Control and Prevention (CDC), National Institute for Occupational Safety and Health (NIOSH) and the Occupational Safety and Health Administration (OSHA) have not established recommended or regulatory occupational exposure limits for DEET.
Please cite as: Jackson, D.; Luukinen, B.; Buhl, K.; Stone, D. 2008. DEET Technical Fact Sheet; National Pesticide Information Center, Oregon State University Extension Services. https://npic.orst.edu/factsheets/archive/DEETtech.html.
References:
- Reregistration Eligibility Decision (RED) DEET; EPA 738-R-98-010; U.S. Environmental Protection Agency, Office of Prevention, Pesticides and Toxic Substances, Office of Pesticide Programs, U.S. Government Printing Office: Washington, DC, 1998; pp 1-34.
- Hazardous Substances Data Bank (HSDB), DEET; HSDB Number 1582; U.S. Department of Health and Human Services, National Institutes of Health, National Library of Medicine. https://toxnet.nlm.nih.gov (accessed Jan 2008), updated March 2003.
- Technical Guide No. 36: Personal Protective Measures Against Insects and Other Arthropods of Military Significance; Defense Pest Management Information Analysis Center (DPMIAC), Armed Forces Pest Management Board (AFPMB): Washington, DC, 2002; pp 21-25.
- Pesticide Products. Pest Bank [CD-ROM] 2007.
- Toxicity and Exposure Assessment for Children's Health (TEACH) Summaries: Diethyltoluamide (DEET) Chemical Summary; U.S. Environmental Protection Agency. https://www.epa.gov/teach/chem_summ/DEET_summary.pdf (accessed Aug 2007), updated April 2007.
- Davis, E. E.; Sokolove, P. G. Lactic Acid-sensitive Receptors on the Antennae of the Mosquito, Aedes aegypti. J. Comp. Physiol. 1976, 105, 43-54.
- McIver, S. B. A model for the mechanism of action of the repellent DEET on the Aedes Aegypti (Diptera: Culicidae). J. Med. Entomol. 1981, 18 (5), 357-361.
- Snow, W. F. The effect of a reduction in expired carbon dioxide on the attractiveness of human subjects to mosquitoes. Bull. Ent. Res. 1970, 60, 43-48.
- Sudakin, D. L.; Trevathan, W. R. DEET: A review and update of safety and risk in the general population. J. Toxicol. Clin. Toxicol. 2003, 41 (6), 831-839.
- Lewis, C. M., Silva, M., Sanborn, J. N,N-Diethyl-Meta-Toluamide (DEET) Risk Characterization Document; California Environmental Protection Agency, Department of Pesticide Regulation: Sacramento, CA, 2000; pp 18-21, 32-33.
- Gwaltney-Brant, S. Insecticides and Molluscicides. Clinical Veterinary Toxicology; Plumlee, K. H., Ed.; Mosby, Inc.: St. Louis, 2004; pp 177-192.
- Bell, J. W.; Veltri, J. C.; Page, 12. B. C. Human Exposures to N,N-diethyl-m-toluamide Insect Repellents Reported to the American Association of Poison Control Centers 1993-1997. Int. J. Toxicol. 2002, 21 (5), 341-352.
- Reigart, J. R.; Roberts, J. R. Other Insecticides, Acaricides, and Repellents. Recognition and Management of Pesticide Poisonings, 5th ed.; U.S. Environmental Protection Agency, Office of Prevention, Pesticides and Toxic Substances, Office of Pesticide Programs, U.S. Government Printing Office: Washington, DC, 1999; pp 80-82.
- Schoenig, G. P.; Osimitz, T. G.; Gabriel, K. L.; Hartnagel, R.; Gill, M. W.; Goldenthal, E. I. Evaluation of the Chronic Toxicity and Oncogenicity of N,N-diethyl-m-toluamide (DEET). Toxicol. Sci. 1999, 47 (1), 99-109.
- Schoenig, G. P.; Osimitz, T. G. DEET. Handbook of Pesticide Toxicology, 2nd ed.; Krieger, R. I., Ed.; Academic Press, Inc.: San Diego, 2001; Vol. 2, pp 1439-1455.
- Knowles, C. O. Miscellaneous Pesticides. Handbook of Pesticide Toxicology; Hayes Jr., W. J., Laws Jr., E.R., Eds.; Academic Press, Inc.: San Diego, 1991; Vol. 3, pp 1500-1505.
- Ross, E. A.; Savage, K. A.; Utley, L. J.; Tebbett, I. R. Insect repellent interactions: sunscreens enhance DEET (N,N-diethyl-mtoluamide) absorption. Drug Metab. Dispos. 2004, 32 (8), 783-785.
- Kasichayanula, S.; House, J. D.; Wang, T.; Gu, X. Percutaneous characterization of the insect repellent DEET and the sunscreen oxybenzone from topical skin application. Toxicol. Appl. Pharmacol. 2007, 223 (2), 187-194.
- Selim, S.; Hartnagel, R. E., Jr.; Osimitz, T. G.; Gabriel, K. L.; Schoenig, G. P. Absorption, Metabolism, and Excretion of N,N,- Dietyl-m-toluamide Following Dermal Application to Human Volunteers. Environ. Appli. Toxicol. 1995, 25, 95-100.
- Stinecipher, J.; Shah, J. Percutaneous Permeation of N,N-Diethyl-m-toluamide (DEET) From Commercial Mosquito Repellents and the Effect of Solvent. J. Toxicol. Environ. Health 1997, 52, 119-135.
- Brand, R. M.; Jendrzejewski, J. L.; Henery, E. M.; Charron, A. R. A Single Oral Dose of Ethanol Can Alter Transdermal Absorption of Topically Applied Chemicals in Rats. Toxicol. Sci. 2006, 92 (2), 349-355.
- Usmani, K. A.; Rose, R. L.; Goldstein, J. A.; Taylor, W. G.; Brimfield, A. A.; Hodgson, E. In Vitro Human Metabolism and Interactions of Repellent N,N-Diethyl-m-toluamide. Drug Metab. Dispos. 2002, 30 (3), 289-294.
- Snodgrass, H. L.; Nelson, D. C.; Weeks, M. H. Dermal penetration and potential for placental transfer of the insect repellent, N,N-diethyl-m-toluamide. Am. Ind. Hyg. Assoc. J. 1982, 43 (10), 747-753.
- Schoenig, G. P.; Hartnagel, R. E., Jr.; Osimitz, T. G.; Llanso, S. Absorption, distribution, metabolism, and excretion of N,Ndiethyl- M-toluamide in the rat. Drug Metab. Dispos. 1996, 24 (2), 156-163.
- CDC. Third National Report on Human Exposure to Environmental Chemicals; U.S. Department of Health and Human Services, Centers for Disease Control and Prevention: Atlanta, 2005.
- Rivera-Cancel, G.; Bocioaga, D.; Hay, A. G. Bacterial Degradation of N,N-Diethyl-m-Toluamide (DEET): Cloning and Heterologous Expression of DEET Hydrolase. Appl. Environ. Microbiol. 2007, 73 (9), 3105-3108.
- Seo, J.; Lee, Y. G.; Kim, S. D.; Cha, C. J.; Ahn, J. H.; Hur, H. G. Biodegradation of the Insecticide N,N-Diethyl-m-Toluamide by Fungi: Identification and Toxicity of Metabolites. Arch. Environ. Contam. Toxicol. 2005, 48 (3), 323-328.
- Sandstrom, M. W.; Kolpin, D. W.; Thurman, E. M.; Zaugg, S. D. Widespread detection of N,N-diethyl-m-toluamide in U.S. streams: comparison with concentrations of pesticides, personal care products, and other organic wastewater compounds. Environ. Toxicol. Chem. 2005, 24 (5), 1029-1034.

